locations in rice (Oryza sativa L.)
G.B. PRASHANTH, S. Hitalmani, SRINIVASACHARY, K. SHADAKSHARI
and H.E. SHASHIDHAR
Department of Genetics and Plant Breeding, University of
Agricultural Sciences, GKVK, Bangalore-560
Blast disease caused by Pyricularia grisea Sacc. is the
most serious fungal disease of rice (Oryza sativa L.). One hundred and
fourteen doubled haploid (DH) lines of a cross between 1R64, an indica
variety adapted to irrigated conditions and Azucena, an upland aromatic
japonica variety (Guiderdoni et a!. 1992) were used for molecular mapping
of QTL associated with leaf and neck blast resistance. The RFLP map and
molecular data generated by Huang eta!. (1994) were used for QTL mapping.
The DH population, along with their parents and susceptible checks 1R50,
HR12 and C039 were screened for blast at IRRI, Philippines in 1997 and
at three locations in South India (1995) in Uniform Blast Nursery. Leaf
blast was scored using 0-5 scale (Mackill and Bonman, 1992) at regular
intervals from seventh day after inoculation to transplanting date. Neck
blast was scored prior to harvest (Anonymous, 1996) in the main field.
Three traits, i.e., percentage diseased leaf area (DLA%), number of susceptible
lesions (LSN) and susceptible lesion size (LSI) were scored in all the
four selected locations.
The phenotypic evaluation for blast indicated IR64 to posses
high level of leaf blast resistance across four locations. The male parent
Azucena was moderately susceptible in all the locations. Nursery evaluation
at IRRI, Philippines showed thirty two partially resistant DH lines whereas
nineteen exhibited resistance in single spore inoculation using P06-06
isolate. Nine doubled haploid lines and IR64 showed resistant lesions in
the presence of high level of inoculum in nursery at Ponnampet (1995),
South India. Ghesquiere et al. (1996) in their study with the same population
identified few resistant lines in France using six diverse strains of Magnaporthe
grisea. Sanger eta!. (1997) observed resistant lesions on IR64 in highly
favorable blast disease environment of the Northern Hill zone of Chhatisgarh,
Madhya Pradesh, India.
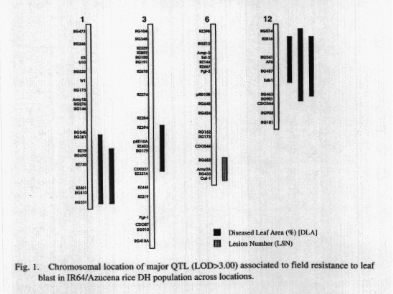
Interval mapping of QTL using the computer package MAPMAKER/QTL
(Lincoln eta!. 1992) with threshold LOD »= 2.00, identified a total
of twenty five QTL for leaf blast and two QTL for neck blast resistance.
Among them, fifteen QTL were detected at blast nursery, IRRI and ten at
Ponnampet. Between the two QTL identified for neck blast resistance, Bangalore
and Mudigere contributed one QTL each (Table 1). Seventeen QTL were identified
for DLA (qDLA), six QTL for LSN (qLSN) and two QTL for LSI (qLSI). Eight
QTL had major effect (LOD »= 3.00) for leaf blast resistance (Table
1, Fig. 1). Some of the QTL were expressed in more than one location and
are presented in Table 2, and multiple effect of chromosomal segment was
hypothesized for these QTL. These results insinuate that pleiotropism rather
than close linkage of different QTL could be the major reason why QTL for
different blast related traits were frequently detected in the same intervals
across locations. QTL on chro.# 9 controlling neck blast resistance (qNBL)
in Bangalore (LOD = 2.03, 8.5% variation) overlapped leaf blast resistance
QTL flanked by RG358-RZ12 (LOD = 2.76, 6.00% variation) of CO39fMoroberekan
recombinant inbred population screened at Ponnampet. The minor QTL for
lesion size on chro.#1 1 bracketed by RG1O3-Npb186 flanking markers (LOD
= 2.02, 9.50% variation) partially overlapped major gene Pi-7(t) (Wang
et a!., 1994) linked to RG1O3A-RG16 markers in Moroberekan, a resistant
japonica variety. The presence of QTL in one location and it�s absence
in another indicated the differences in the pathogenic races. While, the
major QTL identified across wide geographic locations and that too on similar
chromosomal location indicates the stability of the QTL for blast resistance.
Such markers associated with QTL which are common across locations can
be used for selecting blast resistant lines. Our study will give reliable
data on stability of the molecular markers to be used in Marker-assisted
selection, map based cloning and developing of isogenic lines for blast
disease resistance.
Table I. QTL identified for leaf blast resistance in 1R64/Azucena
DH population across locations
DLA�95 = Disease Leaf Area recorded durign 1995. LSN = Number
of susceptible lesions. DLA�96 = Disease Leaf Area recorded during 1996.
NBL = Neck Blast.
|
||||||||||||||||||||||||||||||||||||||||||||
|
* = QTL nomenclature is according to McCourch et a!. (1997).
|
|
Table 2. Multiple Effect of QTL identified across locations
(Threshold LOD>2.00)
* = QTL numenclature is according to McCourch et al. (1997).
Anonymous, 1996. Standard evaluation system for rice (4th
edition). IRRI, P.O.Box 933, Los Banos, Philippines. ppl7-l9.
Ghesquiere, A., M. Lorleux, E. Rournen, L. Albar, N., Huang
and J.L. Nottenghem, 1966. Indica/japonica doubled haploid population as
a model for mapping rice yellow mottle virus and blast resistance genes.
IRRN 21(2-3): 47-49.
Guiderdoni, E., E. Gallinato, J. Luistro and G. Vergara,
1992. Anther culture of tropical japonica/indica hybrids of rice (Oryza
sativa L.). Euphytica 62: 2 19-224.
|
|
Sanger,
|
R.B.S., K.C. Agarwal, M.N. Srivastva andA.K. Sarangi, 1997.
Reaction of promising rice genotypes
|
|
Wang,
|
O.L., D.J. Mackill, M. Bonman, S.R. McCouch, M.C. Champoux
and R.J. Nelson, 1994. RFLP map
|