| 20. Starch degradation during
gibberellin-induced leaf sheath growth in rice
C. MATSUKURA and J. YAMAGUCHI
Bioscience Center and Graduate School of Bioagricultural
Sciences, Nagoya University, Chikusa-ku, Nagoya, 464-8601 Japan
Gibberellins (GAs) promote elongation of rice leaf sheath
with degradation of starch granules in parenchyma cells (Matsukura et a!.
1998). Although some physiological aspects of this phenomena have been
reported, regulatory mechanism(s) of the starch degradation in the leaf
cells remain obscure and further analyses is needed. For this purpose we
examined the expression and the activity of starch phosphorylase which
catalyzes the a-1,4 bonds of starch molecules and releases glucose-l-phosphate.
The plant materials were prepared by a modification of the
�microdrop method�. The seedlings were pretreated with a GA-biosynthesis
inhibitor, uniconazole, to reduce the endogenous GA level and to enhance
the sensitivity to exogenous GAs (Matsukura et a!. 1998). Seedlings at
0 - 48 h after application of GA3 at 300 pmol/plant were excised and fixed
with FAA solution (5% formaldehyde, 5% acetic acid and 45% ethanol), dehydrated
through tert-butyl alcohol series, embedded in Paraplast plus tissue embedding
medium (Sherwood Medical, St. Louis, Mo., USA), sectioned longitudinally
at 10-12 um with a rotary microtome. The sections were stained to visualize
starch granules by PAS (Periodic acid and shift) staining. After application
of GA3, number of starch granules in the leaf sheath parenchyma cells decreased
rapidly, whereas dense granules remained in the control plants (Fig. 1).
Under these conditions the time-course change of phosphory lase activity
during the leaf sheath growth was measured. Fresh rice leaf sheath were
ground with a three-fold amount (w/v) of extraction buffer consisting of
100 mM HepesNaOH (pH 7.5), 1 mM EDTA, 5 mM MgCl2. The homogenate was centrifuged
at 10,000 x g for 10 min at 4°C. The supernatant was assayed for starch
phosphorylase directly or after boiling, and later was used as control
to determine the endogenous glucose-i -phosphate level. Phosphorolytic
activity was measured in coupled enzyme assay as described by Preiss et
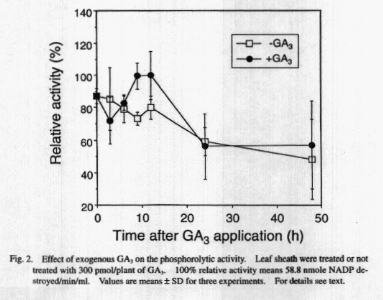
al. (1980). As shown in Fig. 2, phosphorolytic activity of GA3-applied
leaf sheath distinctively increased than that of untreated control at 9
h to 12 h when the striking starch degradation occurred (Fig. 1), indicating
a positive relationship between promotion of the starch degradation and
increase in the phosphorolytic activity (probably an enzyme activation).
We obtained and cloned two partial cDNAs encoding starch
phosphorylase from rice genome project, one (accession number; D23280)
is highly homologous to type L isozyme and another (D46277) to type H isozyme
of potato starch phosphorylase, respectively. It has been known that type
L isozyme is localized within plastids and has a low affinity to oligoglucans,
whereas the type H isozyme is located within the cytosol and has a high
affinity for oligoglucans (Conrads et a!. 1986). The accumulation of starch
phosphorylane mRNA during the growth was detennined by northern blot experiments
using above two clones as probe. No significant differences were observed
in the size or accumulation levels of both mRNAs through the time-course,
and also no difference of plastidic phosphorylase protein using the type
L isozyme specific antibody of potato (data not shown). However we cannot
rule out the possibility that total phosphorylase activity shown in Fig.
2 was due to the expression level of type H isozyme. Recently, it was reported
that a redox potential generated by photosynthesis is involved in the activation
of plastidic acetyl-CoA carboxylase (ACCase) which catalyzes a first step
of fatty acid synthesis in chloroplasts (Sasaki eta!. 1997). It is likely
that gibberellins affect the phosphorylase activities through modulation
of the redox potential and cytosolic and/or plastidic pH, resulting in
the striking degradation of starch granules in leaf sheath cells.
|